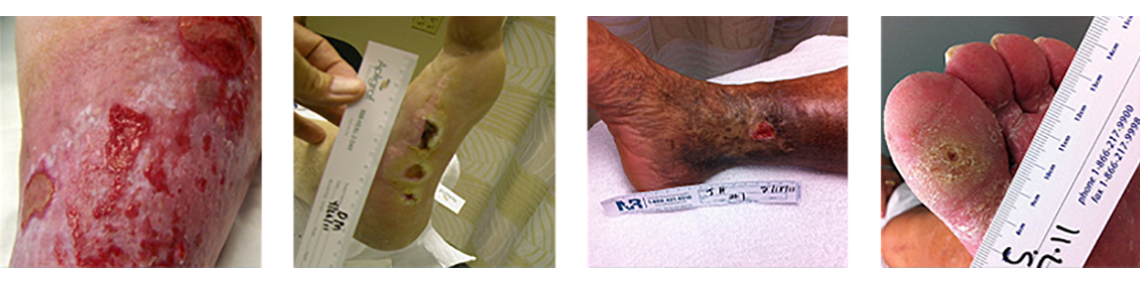
Chronic Wounds Treated with Talymed® Showed Superior Healing Rates1
Talymed® advanced sterile wound matrix is composed of shortened fibers of poly-N-acetyl glucosamine (pGlcNAc), isolated from microalgae.
Proven Efficacy
In a randomized, multi-center clinical trial, significantly more (86.4% vs. 45%) VLU patients experienced complete wound healing in 20 weeks when receiving standard of care (multilayer compression) plus Talymed vs. standard of care alone.1

-
Convenient and Easy to Use
- No prep or mixing time
- Stable at room temperature
- Integrates into scaffold bed
- Fewer dressing changes
Safety
- No known contraindications
- Non-immunogenic
Effectively Manages a Wide Range of Wounds
- Talymed is indicated for the management of a range of serious, complex wounds including:
- Diabetic ulcers
- Venous ulcers
- Pressure wounds
- Dehisced surgical wounds
- Ulcers caused by mixed vascular etiologies
- Full-thickness and partial-thickness wounds
- Second degree burns
- Surgical wounds/donor sites/grafts
- Post-Mohs surgery
- Post-laser surgery
- Abrasions, lacerations
- Traumatic wounds healing by secondary intention
- Chronic vascular ulcers
To learn more about Talymed's superior efficacy, contact us today.
Indications for Use:
Talymed® is indicated for the management of wounds including: diabetic ulcers, venous ulcers, pressure wounds, ulcers caused by mixed vascular etiologies, full thickness and partial thickness wounds, second degree burns, surgical wounds-donor sites/grafts, post-mohs surgery, post-laser surgery, and other bleeding surface wounds, abrasions, lacerations, traumatic wounds healing by secondary intention, chronic vascular ulcers, dehisced surgical wounds.
Safety Information:
PRECAUTIONS: There are no known contraindications to the use of Talymed®.
CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician or other licensed practitioner.
Manufactured by Marine Polymer Technologies, Inc. For instructions for use or additional product information, please contact us.
- 9,139,664
- 9,139,663
- 8,871,247
Talymed® - Ordering Information:
| Item # | Description | Matrix Size |
|---|---|---|
| 400-20 | Talymed 5 cm x 5 cm | 25 sq cm |
| 400-35 | Talymed 3 cm x 3 cm | 9 sq cm |
